Research topics
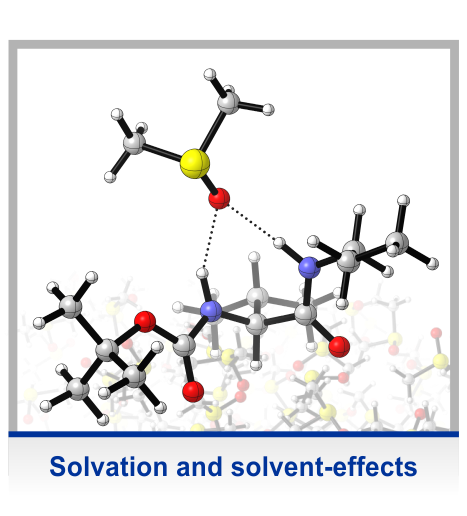
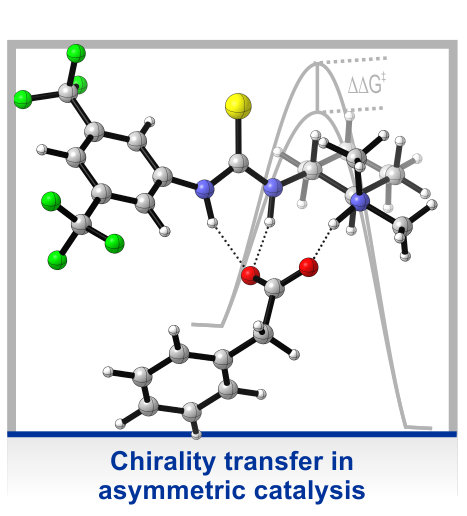
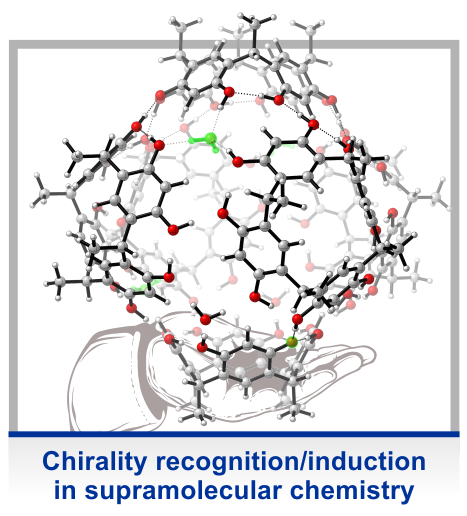
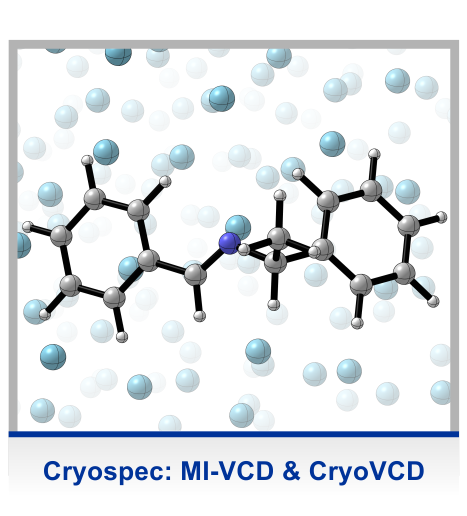
In many of our projects, molecular chirality is an important intrinsic property of the system under study. This is the case, for instance, when studying active conformations of asymmetric catalysts, i.e. the structure of the complex of a reactant and the catalysts. However, especially in solvation-related projects, we often use simple chiral model systems, where the chirality is introduced solely to take advantage of the structural sensitivity of vibrational circular dichroism (VCD) spectroscopy and to probe changes in molecular conformation in high detail. These studies enabled us to establish the concept of using vibrational optical activity as probe for intermolecular interactions.
Vibrational CD spectroscopy
The main experimental technique applied in our studies is VCD spectroscopy, most likely the most powerful chiroptical spectroscopic technique. It can be regarded as the extension of the well-known electronic CD spectroscopy to the infrared region and as such it measures the very small differential absorption of left- and right-circular polarized light during a vibrational transition (VCD = A_LCP – A_RCP). Although the effect is about four orders of magnitude smaller than the actual vibrational absorbance, it is much more sensitive to conformational changes due to interactions with solvent or other molecules in solution. The interpretation of a VCD spectrum needs extensive density functional theory (DFT) based spectra calculations and a comprehensive conformational sampling.

Absolute Configurations
The key application of VCD spectroscopy is the determination of absolute configurations of chiral molecules. The VCD spectrum of a given enantiomer of a chiral molecule shows either a positive or a negative band for each of the vibrational modes, depending on the preferential absorbance of either type of circularly polarized light. Similarly, the opposite enantiomer will feature a mirror imaged spectrum with all bands having opposite signs. Therefore, by comparison with computationally predicted IR and VCD spectra, the spectral signatures can easily be used to assign the AC of the sample with very high confidence. In a recent perspective article in J. Org. Chem we show how such an AC determination is carried out.


Feel free to contact Christian in case you are interested in AC determinations of your samples.